Blog
IEC 62304 for Medical Device Software: Requirements, Classes & Lifecycle

Medical device software is no longer a supporting component; it is often the core of diagnosis, monitoring, and therapy. As software directly influences patient outcomes, regulators expect manufacturers to prove that it is designed, developed, and maintained under a disciplined, risk-based lifecycle. IEC 62304 sits at the center of that expectation, defining how medical device software must be planned, built, tested, released, and maintained to protect patient safety and device effectiveness.
IEC 62304 compliance is not about rigid documentation or a prescribed development model. It is about demonstrating control: clear requirements, traceability from risk to code to test, proportional rigor based on software safety classification, and objective evidence that failures are anticipated and managed.
Whether software is embedded in a physical device or delivered as Software as a Medical Device (SaMD), IEC 62304 provides the framework regulators rely on to evaluate software quality, safety, and reliability, making it a foundational standard for global market access.
IEC 62304 Essential Overview
IEC 62304 is the internationally recognized standard that defines software life cycle processes for medical device software. It establishes what manufacturers must do to ensure software safety and effectiveness, without prescribing a specific development methodology.
The standard applies a risk-based approach, meaning the higher the potential patient harm, the stricter the development and verification expectations.
At its core, IEC 62304 ensures that medical device software is:
- Developed in a controlled and repeatable manner
- Traceable from requirements through testing
- Risk-informed, aligned with patient safety goals
- Maintainable throughout its operational life
What Is IEC 62304?
IEC 62304 is a software life cycle standard published by the International Electrotechnical Commission (IEC). It specifies the processes, activities, and tasks required to develop and maintain software used in medical devices safely.
Unlike product standards, IEC 62304 does not evaluate whether a device is clinically effective or fit for a specific medical purpose. Instead, it focuses on how software is engineered, ensuring that failures, anomalies, and changes are systematically controlled throughout the software’s life cycle.
The standard applies to:
- Embedded software that controls or monitors a medical device
- Standalone software that qualifies as Software as a Medical Device (SaMD)
Importantly, IEC 62304 is technology-agnostic. Whether the software runs on mobile, cloud, desktop, or proprietary hardware platforms, its obligations remain the same if the intended use qualifies it as medical device software.
What Does 62304 Compliance Mean?
62304 compliance does not mean following a specific template, tool, or development model. It means being able to demonstrate control over the software life cycle objectively.
In practice, IEC 62304 compliance requires evidence that:
- Software development activities are planned and defined
- Software requirements are documented, testable, and traceable
- Risks related to software failure are identified, evaluated, and controlled
- Verification and testing activities are appropriate to the software safety class
- Changes and problems are managed under configuration and problem resolution processes
The standard deliberately avoids mandating document formats. Evidence may exist in formal documents, configuration-managed repositories, ticketing systems, or validated development tools, as long as traceability and control can be demonstrated.
IEC 62304 Standard vs IEC 62304 Software
IEC 62304 is often misunderstood as a “software quality” or “testing” standard. In reality, it is a software life cycle process standard.
It does not:
- Define clinical performance requirements
- Replace system-level validation or usability engineering
- Prescribe coding languages, tools, or architectures
Instead, IEC 62304 defines how software-related work must be organized, reviewed, verified, and maintained so that software failures do not introduce unacceptable risk to patients or users.
This distinction is critical during audits: regulators assess whether lifecycle activities exist, are appropriate for the risk class, and are consistently applied, not whether a specific development method was used.
IEC 62304 Medical Device Software Standard: Scope and Applicability
Medical device software is defined by intended use, not by where or how it runs. Software is considered medical device software if it performs a medical function such as diagnosis, monitoring, treatment, or therapy support.
IEC 62304 applies equally to:
- Embedded software inside physical devices (e.g., infusion pumps, imaging systems)
- Software as a Medical Device (SaMD) running on general-purpose platforms (e.g., mobile apps, cloud-based diagnostic software)
Changing the deployment platform does not change IEC 62304 obligations if the medical intent remains the same.
ISO 62304 vs IEC 62304: Clearing the Confusion
“ISO 62304” is a commonly searched term, but the correct standard is IEC 62304. While ISO and IEC collaborate closely and distribute standards globally, IEC 62304 remains an IEC-issued standard. Regulatory authorities and notified bodies reference IEC 62304 when evaluating medical device software lifecycle compliance.
IEC 62304 Software Requirements for Medical Devices
Software requirements are foundational to IEC 62304 compliance. They define what the software must do, how it must perform, and how it supports risk control.
Under IEC 62304, software requirements must be:
- Unambiguous and understandable
- Traceable to system requirements and risk control measures
- Testable, with clear acceptance criteria
- Maintained throughout the software life cycle
Security-related requirements, such as data integrity, availability, and access control, are also expected to be included where relevant, even though IEC 62304 itself does not deeply prescribe cybersecurity techniques.
IEC 62304 Software Life Cycle for Medical Devices
IEC 62304 does not mandate a waterfall or V-model lifecycle. Instead, it requires that specific tasks and outcomes are achieved across the life cycle, regardless of the development approach used.
These lifecycle expectations include:
- Structured planning
- Controlled requirements and design
- Risk-informed development and testing
- Formal release decisions
- Ongoing maintenance and problem resolution
Agile, hybrid, and iterative development models can all meet IEC 62304 requirements, provided the required activities are performed and evidence is maintained.
IEC 62304 Software Safety Classification
The central principle of IEC 62304 is risk-based rigor. The standard assumes that software can fail at any time and adjusts development expectations based on the severity of potential harm that a software failure could cause. This approach is known as software safety classification.
IEC 62304 defines three software safety classes: A, B, and C. The higher the class, the more stringent the lifecycle, verification, and documentation requirements.
IEC 62304 Software Safety Classes
| Software Class | Potential Harm | Practical Meaning |
| Class A | No injury or damage to health is possible | Failures do not result in patient harm |
| Class B | Non-serious injury is possible | Failures may cause temporary or reversible injury |
| Class C | Death or serious injury is possible | Failures could be life-threatening or permanently harmful |
The classification is determined by evaluating worst-case failure scenarios, not intended benefits or normal operating conditions.
What Are the IEC 62304 Class A Requirements?
IEC 62304 Class A software represents the lowest risk category, but it is not exempt from lifecycle controls. Class A software must still demonstrate structured development, controlled requirements, and appropriate verification activities.
Key characteristics of Class A compliance include:
- Defined software development planning
- Documented software requirements
- Architecture and design evidence (lighter detail than B/C)
- System-level testing covering all requirements
- Configuration and problem resolution processes
While Class A does not require the same depth of unit verification or detailed design documentation as higher classes, manufacturers must still prove that software failures cannot result in patient harm.
What Is IEC 62304 Class C?
IEC 62304 Class C software represents the highest risk category. If a software failure could contribute to death or serious injury, the software must be treated as Class C.
Class C compliance requires:
- Detailed architectural and unit-level design documentation
- Formal unit implementation and verification
- Robust integration and system testing
- Strong traceability between risks, requirements, design, and tests
- Controlled release decisions with safety justification
Regulators expect Class C software to show a high level of engineering discipline and objective evidence that safety risks have been reduced to acceptable levels.
Common Software Safety Classification Mistakes
Several misunderstandings frequently lead to incorrect classification:
Software benefits do not lower classification
Classification is based on potential harm if the software fails, not on how beneficial the software is when it works correctly.
Risk controls implemented in software do not reduce classification
Even if safeguards are implemented within the software, failures must still be assumed possible. Software-based controls do not justify down-classifying risk.
Assuming the software failure probability is low
For classification purposes, IEC 62304 assumes software can fail at any time. Probability arguments alone cannot justify a lower safety class.
Correct classification is foundational. An incorrect safety class can invalidate the entire compliance strategy.
IEC 62304 Compliance: Core Lifecycle Processes
IEC 62304 organizes software activities into five core process groups. Together, these ensure that software is developed, maintained, and corrected under controlled conditions.
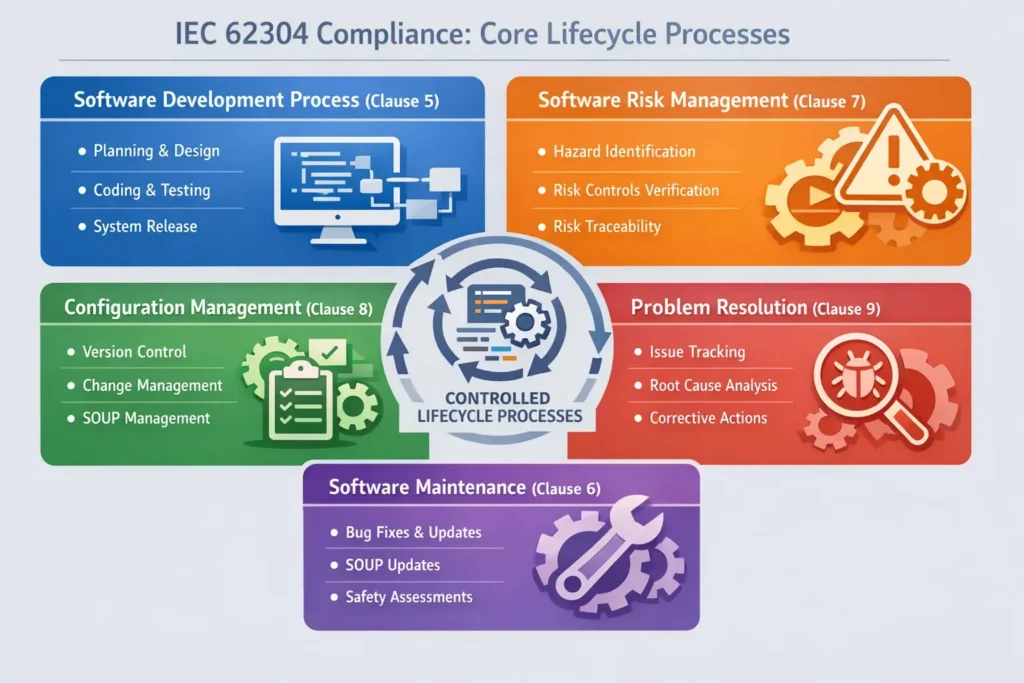
Software Development Process (Clause 5)
The software development process defines how software is planned, designed, implemented, tested, and released. The rigor of each activity scales with the software safety class.
Key development activities include:
- Software development planning
- Software requirements analysis
- Software architectural design
- Software detailed design
- Software unit implementation and verification
- Software integration and integration testing
- Software system testing
- Software release
These activities do not need to follow a strict sequence, but they must all be performed and documented.
Software Risk Management Process (Clause 7)
IEC 62304 integrates closely with medical device risk management practices. Software risk management focuses on how software failures can contribute to hazardous situations identified at the system level.
Key expectations include:
- Identifying software-related causes of hazards
- Linking software requirements to risk control measures
- Verifying that software risk controls are effective
- Maintaining traceability between risks, requirements, and verification
Risk management is not a one-time activity. It must evolve as the software changes throughout its life cycle.
Software Configuration Management Process (Clause 8)
Configuration management ensures that software and its related artifacts remain identifiable, traceable, and controlled.
This includes:
- Version control of source code and documentation
- Defined baselines for releases
- Controlled change processes
- Identification and management of SOUP (Software of Unknown Provenance)
Without strong configuration management, it becomes impossible to prove which version of the software was tested or released.
Software Problem Resolution Process (Clause 9)
Problem resolution ensures that anomalies, bugs, and failures are handled systematically.
Under IEC 62304, manufacturers must:
- Record and evaluate software problems
- Assess safety impact
- Investigate root causes
- Implement and verify corrective actions
- Track issues to closure
Problem resolution applies during development and after release, reinforcing the idea that software safety is a continuous responsibility.
Software Maintenance Process (Clause 6)
Once software is released, IEC 62304 requires ongoing control through a defined maintenance process.
Maintenance activities address:
- Bug fixes and enhancements
- Changes triggered by risk analysis
- Updates to third-party or SOUP components
- Post-market safety information
All changes must be evaluated for their impact on safety and verified appropriately.
IEC 62304 Versions and Updates
Understanding which version of IEC 62304 applies is essential for regulatory alignment and audit readiness. While multiple dates are referenced online, they do not represent entirely separate standards; they reflect updates to the same core framework.
IEC 62304:2006
IEC 62304 was first published in 2006 to address the growing role of software in medical devices. This initial edition established the foundational software lifecycle processes that are still in use today, including development, maintenance, risk management, configuration management, and problem resolution.
IEC 62304:2015 (Amendment 1)
The version most commonly referenced today is IEC 62304:2006 + Amendment 1 (2015). Rather than introducing a new edition, the 2015 amendment clarified and refined existing requirements, particularly around risk management alignment and lifecycle consistency.
This consolidated version is often informally referred to as IEC 62304:2015, even though the base standard remains the 2006 edition.
Which Version of IEC 62304 Is Currently Valid?
For practical and regulatory purposes, the consolidated IEC 62304:2006 + A1:2015 is the version currently relied upon by manufacturers, notified bodies, and regulators.
It is also the version recognized by major regulatory authorities, including the U.S. FDA, as a consensus standard for medical device software lifecycle processes.
IEC 62304 Latest Version: What Teams Should Use Today
Until a formal second edition is published, organizations should:
- Align software lifecycle processes with IEC 62304:2006 + A1:2015
- Ensure risk management alignment with ISO 14971
- Maintain objective evidence that lifecycle activities are consistently applied
This approach remains accepted globally for both legacy and new software development projects.
IEC 62304 vs Other Standards
IEC 62304 does not exist in isolation. It operates within a broader ecosystem of medical device and software standards, each serving a distinct purpose.
IEC 62304 vs ISO 62304
There is no separate “ISO 62304” standard. IEC 62304 is authored and maintained by the International Electrotechnical Commission (IEC). While ISO and IEC collaborate closely, regulatory bodies and auditors explicitly reference IEC 62304 for medical device software lifecycle compliance.
IEC 62304 vs IEC 62443
IEC 62304 and IEC 62443 serve fundamentally different domains:
- IEC 62304 focuses on software lifecycle processes for medical device software, emphasizing patient safety and risk-based development.
- IEC 62443 addresses cybersecurity for industrial automation and control systems, such as manufacturing plants and critical infrastructure
While cybersecurity considerations may intersect with medical software development, IEC 62443 is not a replacement for IEC 62304 and is generally not applied directly to medical device software compliance.
Relationship with ISO 13485 and ISO 14971
IEC 62304 complements, rather than replaces, other core medical device standards:
- ISO 13485 defines the quality management system requirements for medical device manufacturers.
- ISO 14971 defines the risk management framework across the entire device lifecycle.
- IEC 62304 operationalizes software-specific lifecycle and risk activities within that framework.
Together, these standards form the backbone of regulatory expectations for software-enabled medical devices.
Agile + IEC 62304: Can They Work Together?
IEC 62304 does not prescribe a development methodology, which makes Agile development fully compatible when implemented correctly.
The standard explicitly allows manufacturers to select their own lifecycle model, provided all required tasks and activities are performed and documented.

Making Agile Development IEC 62304-Compliant
Agile teams commonly meet IEC 62304 requirements by mapping existing practices to lifecycle expectations:
- User stories and backlog items serve as software requirements
- Acceptance criteria support verification planning
- Continuous integration pipelines provide repeatable verification evidence
- Sprint reviews and retrospectives support problem resolution and change control
- Version control systems support configuration management
The key requirement is not the format of the artifacts, but the ability to demonstrate traceability, control, and risk awareness.
What Auditors Expect to See in Agile Teams
Auditors typically look for:
- A documented explanation of how Agile practices map to IEC 62304 tasks
- Clear traceability from risk to requirement to test
- Controlled releases and baselined artifacts
- Evidence that safety-related changes are reviewed and verified
When Agile is implemented with discipline, it often results in stronger, more transparent compliance than rigid document-heavy approaches.
What to Expect in the Next Revision of IEC 62304
A second edition of IEC 62304 is under development, reflecting shifts in how medical and health software is built and deployed.
Expected directions include:
- Broader coverage of health software, not only traditional medical devices
- Simplification of safety classification into two rigor levels
- Stronger alignment with modern software development practices
- Increased emphasis on lifecycle integration rather than isolated activities
While the core principles of risk-based development and traceability will remain unchanged, future revisions are expected to better accommodate cloud-based software, frequent updates, and modern delivery models.
Is IEC 62304 Mandatory?
IEC 62304 is not a law by itself. However, it represents the state of the art for medical device software lifecycle management.
Manufacturers that choose not to follow IEC 62304 are expected to justify how their alternative approach provides an equivalent level of safety and control, a position that is difficult to defend during regulatory review.
As a result, IEC 62304 is widely treated as a de facto requirement for medical device software development.
Does the FDA Recognize IEC 62304?
Yes. The U.S. FDA recognizes IEC 62304 (consolidated 2006 + A1:2015) as a consensus standard for medical device software lifecycle processes.
Declaring conformity to IEC 62304 can:
- Reduce the need for extensive custom explanations in submissions
- Align software documentation with FDA expectations
- Streamline premarket review of software-related evidence
This recognition reinforces IEC 62304’s role as a foundational standard for global regulatory acceptance.
Turning IEC 62304 Into a Competitive Advantage
IEC 62304 is often treated as a regulatory hurdle, but when implemented correctly, it becomes a force multiplier for medical device software teams. Clear lifecycle control reduces rework, disciplined risk management prevents late-stage surprises, and strong traceability shortens audit cycles and regulatory reviews.
Products developed under a well-implemented IEC 62304 framework are easier to maintain, easier to scale, and significantly more resilient to change, whether driven by clinical feedback, cybersecurity needs, or evolving regulations.
Organizations that succeed with IEC 62304 typically share a few traits:
- Software lifecycle processes are embedded into daily engineering work, not bolted on at release.
- Risk management is treated as a continuous design input, not a compliance checkbox.
- Documentation and traceability are byproducts of good systems, not manual afterthoughts.
- Agile speed is preserved while maintaining regulatory-grade control.
As medical software continues to move toward cloud platforms, frequent updates, AI-enabled functionality, and connected ecosystems, these capabilities are no longer optional. IEC 62304 provides the structure that allows innovation to move quickly, without compromising patient safety or regulatory confidence.
Where IEC 62304 Fits in a Modern Medical Software Stack
IEC 62304 works best when it is aligned with:
- A quality management system that supports controlled change and accountability
- A risk management process that connects system hazards to software behavior
- Secure development practices that address confidentiality, integrity, and availability
- Toolchains that support traceability across requirements, code, tests, and releases
When these elements work together, compliance becomes repeatable rather than reactive, and audits become confirmations, not investigations.
Why IEC 62304 Matters Beyond Compliance
IEC 62304 ultimately exists to protect patients, but its impact extends far beyond safety alone. Teams that implement the standard effectively gain predictability in development, clarity in decision-making, and confidence during regulatory interactions. Instead of relying on individual expertise or tribal knowledge, IEC 62304 creates a shared, auditable understanding of how software is built, changed, and maintained.
From a business perspective, this matters because:
- Regulatory delays are reduced when software evidence is complete and traceable
- Maintenance and updates become safer and faster due to controlled change processes
- Scaling teams or onboarding new engineers is easier with defined lifecycle practices
- Long-term product sustainability improves as technical debt and unmanaged risk decrease
In highly regulated markets, the ability to demonstrate control is just as important as innovation itself.
Aligning IEC 62304 With Real-World Development
One of the most practical strengths of IEC 62304 is its flexibility. It does not force organizations into heavyweight documentation or outdated development models. Instead, it rewards intentional engineering, clear requirements, thoughtful design, disciplined testing, and continuous risk awareness.
When lifecycle activities are integrated into modern toolchains and workflows:
- Requirements live close to code and tests
- Traceability is automated rather than manual
- Verification evidence is generated continuously
- Releases are supported by objective, reviewable data
This alignment allows teams to remain fast and adaptive while still meeting regulatory expectations.
Final Perspective
IEC 62304 is not simply a standard to “check off” before submission. It is a framework that defines what responsible medical device software development looks like today and in the future. As software continues to take on a greater clinical role, the principles embedded in IEC 62304 will only become more central to how regulators evaluate safety, effectiveness, and quality.
Implementing IEC 62304 effectively requires more than understanding the standard; it requires engineering execution that aligns software development, risk management, and regulatory expectations. Explore our Healthcare Software Development
Discover how regulated healthcare and medical software is designed, built, and maintained with compliance-first engineering practices.
FAQs
Q: When Was IEC 62304 Released?
Ans: IEC 62304 was first published in 2006. The most commonly used version today includes Amendment 1, published in 2015.
Q: What Are the IEC 62304 Class A Requirements?
Ans: Class A software must still follow defined lifecycle processes, including planning, requirements, testing, configuration management, and problem resolution, but with lighter rigor than higher-risk classes.
Q: What Is IEC 62304 Class C?
Ans: Class C is the highest risk category, applied when software failure could result in death or serious injury. It requires the most stringent development, verification, and documentation controls.
Q: Is IEC 62304 Mandatory?
Ans: IEC 62304 is not legally mandatory, but it is widely expected by regulators and notified bodies as evidence of appropriate software lifecycle control.
Q: Does FDA Recognize IEC 62304?
Ans: Yes. The FDA recognizes IEC 62304 as a consensus standard for medical device software lifecycle processes.
References
Table of Contents
1) IEC 62304 Essential Overview
2) What Is IEC 62304?
3) What Does 62304 Compliance Mean?
4) IEC 62304 Standard vs IEC 62304 Software
5) IEC 62304 Medical Device Software Standard: Scope and Applicability
6) IEC 62304 Software Safety Classification
7) IEC 62304 Compliance: Core Lifecycle Processes
8) IEC 62304 Versions and Updates
9) IEC 62304 vs Other Standards
10) Agile + IEC 62304: Can They Work Together?
11) What to Expect in the Next Revision of IEC 62304
12) Is IEC 62304 Mandatory?
13) Does the FDA Recognize IEC 62304?
14) Final Perspective
Innovate the Future of Health Tech
CitrusBits helps MedTech leaders build smarter apps, connected devices, and XR health solutions that truly make an impact.
