Blog
Diagnostic Medical Device Software Engineering: Building FDA-Ready, Scalable Clinical Systems

Developing diagnostic medical device software is no longer just an engineering challenge; it’s a regulatory, clinical, and architectural problem that directly impacts time to market, FDA approval, and patient outcomes. From real-time data processing and AI-driven diagnostics to IEC 62304 compliance and software validation, modern diagnostic systems demand far more than traditional development approaches.
Yet, most organizations struggle not because they lack engineering talent, but because they lack a regulatory-first architecture and a clear path to building audit-ready, clinically reliable systems.
Let’s break down how to engineer diagnostic software that meets FDA requirements, scales with complex data environments, and accelerates your path from prototype to approved medical product.
What Defines Diagnostic Medical Device Software Engineering?
Diagnostic medical device software engineering operates at the intersection of clinical accuracy, regulatory compliance, and high-performance system architecture. Unlike conventional software development, these systems are responsible for capturing, processing, and interpreting physiological or imaging data that directly influences diagnostic decisions and patient outcomes.
At a systems level, diagnostic software can be categorized into two primary architectures:
Embedded Software (Device-Level)
Firmware and low-level systems are integrated directly into diagnostic hardware (e.g., imaging systems, monitoring devices), where latency, signal integrity, and hardware constraints are critical.
Software as a Medical Device (SaMD)
Standalone platforms that ingest, process, and analyze clinical data, often leveraging cloud infrastructure, AI models, and interoperability layers.
What differentiates diagnostic systems is the requirement for clinical-grade reliability and traceability. Every function, from data acquisition to algorithmic output, must be:
- Deterministic and reproducible
- Fully traceable to requirements and risk controls
- Validated against clinical use cases
This leads to a fundamentally different engineering paradigm:
- Architecture must support real-time diagnostic data processing systems
- Systems must align with SaMD product engineering for diagnostics
- Infrastructure must enable clinical-grade diagnostic software architecture, not consumer-grade scalability patterns
In practice, this means engineering teams must design systems where performance, compliance, and clinical validity are co-dependent, not sequential.
Regulatory-First Architecture: Designing for FDA, IEC 62304, and ISO 14971
In diagnostic software engineering, compliance is not a post-development activity; it is an architectural constraint from day one. Organizations that treat regulatory alignment as an afterthought often face costly rework, delayed submissions, or outright rejection during FDA review.
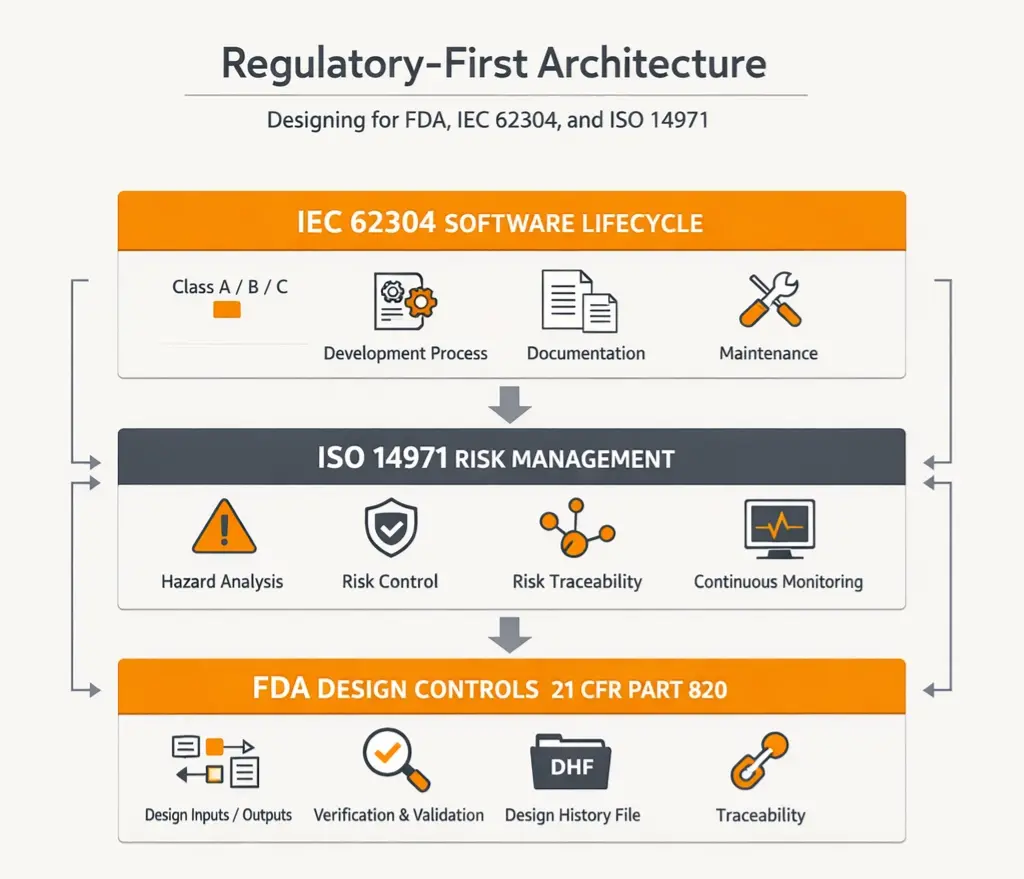
A regulatory-first approach requires aligning system design with three critical frameworks:
1. IEC 62304: Software Lifecycle Standard
Defines the complete lifecycle for medical device software, including:
- Software safety classification (Class A, B, C)
- Development processes and documentation requirements
- Maintenance and post-market updates
This standard enforces structured, auditable development pipelines, making it foundational for any IEC 62304-compliant software engineering services.
2. ISO 14971: Risk Management
Diagnostic systems must continuously evaluate and mitigate risks related to:
- Incorrect diagnostic outputs
- Data corruption or latency
- System failures in real-time environments
This requires implementing:
- Hazard analysis and risk control mechanisms
- Risk traceability across system components
- Continuous risk monitoring throughout the lifecycle
3. FDA Design Controls (21 CFR Part 820)
For FDA Class II medical software development, organizations must establish:
- Design inputs and outputs
- Verification and validation protocols
- Design history files (DHF)
- End-to-end traceability
Architectural Implications
A regulatory-first system is not just documented; it is engineered for auditability:
- Every feature is linked to design controls and risk mitigations
- Code, requirements, and tests are connected through traceability matrices
- Systems are structured to produce audit-ready medical software systems at any stage
This approach fundamentally shifts engineering priorities:
- From speed to controlled, compliant iteration
- From feature delivery to risk-managed system design
- From code quality to clinical and regulatory reliability
Where Most Teams Fail?
The most common failure point is attempting to retrofit compliance into an already built system. This results in:
- Broken traceability
- Incomplete validation artifacts
- Increased regulatory risk
High-performing teams, on the other hand, embed compliance into:
- System architecture
- Development workflows
- Validation pipelines
System Architecture for Diagnostic Platforms: Designing for Real-Time, Clinical-Grade Performance
At the core of every diagnostic medical device lies a data-intensive, latency-sensitive system architecture that must operate with near-perfect reliability. Unlike traditional enterprise systems, diagnostic platforms cannot tolerate delays, inconsistencies, or data loss, because every millisecond and every signal can influence clinical decisions.
The primary architectural challenge is balancing:
- Real-time processing requirements
- Regulatory constraints
- Scalability across devices and environments
Core Architectural Components
1. Real-Time Data Ingestion & Processing
Diagnostic systems rely on continuous streams of physiological or imaging data. This requires:
- High-throughput ingestion pipelines
- Low-latency processing engines
- Deterministic data handling
Modern systems are increasingly built around real-time diagnostic data processing systems that can:
- Process signals at the edge
- Filter and normalize raw inputs
- Deliver near-instant diagnostic outputs
2. Edge vs Cloud Decisioning
A critical architectural decision is where computation happens:
Edge Computing (Device-Level)
- Used for latency-critical diagnostics
- Reduces dependency on connectivity
- Enables immediate clinical feedback
Cloud Infrastructure
- Used for large-scale data aggregation
- AI model training and updates
- Longitudinal patient analysis
High-performance platforms combine both, leveraging edge computing for diagnostic medical devices while maintaining centralized intelligence in the cloud.
3. Interoperability & Clinical Integration
Diagnostic systems must seamlessly integrate into existing healthcare ecosystems. This requires:
- FHIR and HL7-based interoperability layers
- Integration with EHR/EMR systems
- Secure data exchange protocols
Without this, even the most advanced diagnostic platform fails at the point of care.
4. Scalable, Fault-Tolerant Architecture
Clinical environments demand systems that are:
- Highly available
- Fault-tolerant
- Resilient under load
This is where clinical-grade diagnostic software architecture diverges from traditional SaaS:
- Redundancy is mandatory
- Failover mechanisms must be validated
- Downtime is not just a bug; it’s a clinical risk
Where Most Architectures Break
Many organizations attempt to scale diagnostic platforms using generic cloud-native patterns. This leads to:
- Latency issues in real-time diagnostics
- Inconsistent data synchronization
- Regulatory compliance gaps
Diagnostic platforms are not just software systems; they are a real-time clinical infrastructure. Their architecture must be engineered to support accuracy, speed, and compliance simultaneously.
AI in Diagnostic Systems: From Algorithms to FDA-Validated Intelligence
Artificial intelligence is rapidly becoming the backbone of modern diagnostic systems, but integrating AI into medical devices introduces a new layer of complexity that extends far beyond model accuracy.
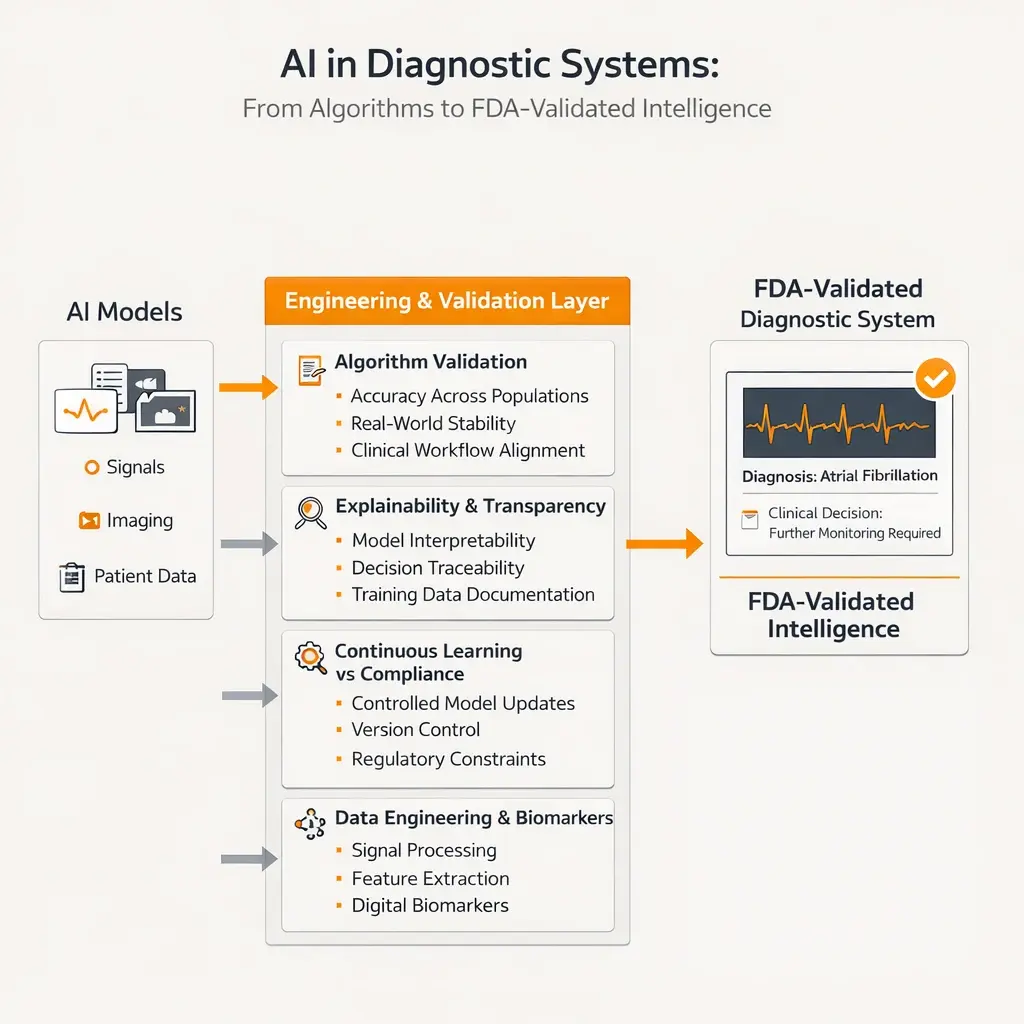
In regulated environments, AI is not just a feature; it is a clinical decision-making component that must be explainable, validated, and continuously monitored.
From Models to Medical-Grade Systems
Most teams focus on building high-performing models. However, in diagnostic systems, success depends on:
- Clinical relevance of outputs
- Reproducibility across datasets
- Regulatory acceptance of algorithms
This requires a shift from “building AI models” to engineering AI diagnostic systems
Key Engineering Challenges
- Algorithm Validation & Clinical Evidence
AI models must undergo rigorous validation to ensure:
- Accuracy across diverse patient populations
- Stability under real-world conditions
- Alignment with clinical workflows
This is where diagnostic algorithm validation becomes critical for FDA submissions.
- Explainability & Transparency
Regulators increasingly require:
- Model interpretability
- Traceable decision logic
- Clear documentation of training data
Black-box models introduce significant regulatory risk, especially in high-risk diagnostic applications.
- Continuous Learning vs Regulatory Constraints
AI systems naturally evolve, but regulated systems must remain stable.
This creates tension between:
- Model improvement
- Regulatory approval constraints
Engineering teams must implement controlled update mechanisms that allow innovation without violating compliance.
- Data Engineering & Digital Biomarkers
Modern diagnostic platforms rely heavily on:
- Signal processing pipelines
- Feature extraction systems
- Digital biomarker identification
This requires advanced capabilities in digital biomarker processing and real-time analytics.
The Biggest Mistake Teams Make
Most organizations treat AI as an add-on rather than a core system component. This leads to:
- Poor integration with clinical workflows
- Validation failures
- Regulatory rejection
AI in diagnostic systems is not about intelligence alone; it’s about engineering trust, validation, and clinical reliability at scale.
Software Verification, Validation (V&V), and Clinical Readiness
In diagnostic medical device software engineering, verification and validation (V&V) is not a phase; it is a continuous, traceable process embedded throughout the lifecycle. This is the point where engineering meets regulatory scrutiny, and where most systems either become FDA-ready or fail under audit.
Verification answers: “Did we build the system right?”
Validation answers: “Did we build the right system for clinical use?”
For diagnostic systems, both must align with clinical intent, risk controls, and regulatory requirements simultaneously.
Components of V&V:
- Multi-Level Testing Strategy
- Unit testing ensures code-level correctness
- Integration testing validates system interactions
- System testing evaluates end-to-end workflows
- Clinical validation confirms real-world diagnostic accuracy
Unlike traditional software, these layers must map directly to design inputs and risk controls.
- Traceability & Documentation
Regulatory bodies require full traceability between:
- Requirements
- Risk controls
- Test cases
- Validation outcomes
This is achieved through software traceability in healthcare systems, often managed via ALM tools and structured documentation.
3. FDA Submission Readiness
For FDA Class II systems, validation must include:
- Documented V&V protocols
- Clinical evaluation reports
- Software validation for FDA submission
- Evidence of risk mitigation
V&V in Diagnostic Software vs Traditional Software
Aspect | Diagnostic Software (Medical) | Traditional Software |
Validation Scope | Clinical + Technical | Functional only |
Traceability | Mandatory (end-to-end) | Rarely enforced |
Risk Alignment | Integrated with ISO 14971 | Minimal |
Documentation | Audit-ready, regulated | Lightweight |
Failure Impact | Patient safety risk | Business/UX impact |
In diagnostic systems, V&V is not just about quality, it is about proving clinical safety and regulatory compliance with evidence.
Hidden Risks in Diagnostic Software Development (Why Most Projects Fail)
Most diagnostic software projects don’t fail because of poor coding, they fail because of misaligned architecture, regulatory blind spots, and underestimated system complexity.
These risks often remain invisible until late-stage validation or regulatory review, where fixing them becomes exponentially expensive.
Compliance as an Afterthought
- Teams build fast without aligning to IEC 62304
- Documentation is incomplete or inconsistent
- Traceability is broken across systems
Result: Regulatory rejection or major delays
Weak System Architecture
- Over-reliance on generic cloud patterns
- Poor handling of real-time diagnostic data
- Lack of fault tolerance in clinical environments
Result: Unstable diagnostic outputs and system failures
Inadequate Validation Strategy
- Testing limited to functional scenarios
- No clinical validation pipeline
- Missing risk-based test coverage
Result: Unproven system reliability in real-world use
Data & AI Risks
- Biased or insufficient training datasets
- Lack of explainability in diagnostic models
- Poor integration with clinical workflows
Result: Loss of trust from regulators and clinicians
Risk Impact vs Business Outcome
Risk Type | Impact on Product | Business Consequence |
Compliance Gaps | Failed audits | Delayed FDA approval |
Architecture Issues | System instability | Increased rework cost |
Validation Failures | Clinical unreliability | Market rejection |
AI Misalignment | Inaccurate diagnostics | Regulatory risk |
The real cost of diagnostic software failure is not technical, it is regulatory delay, lost market opportunity, and clinical risk exposure.
Build vs Partner: When to Outsource Diagnostic Software Engineering
For many MedTech organizations, the decision is not whether to build diagnostic software, but how to build it without introducing regulatory risk or delaying time to market.
While in-house teams bring product knowledge, they often lack the specialized regulatory and system-level expertise required for diagnostic platforms.
In-House Approach
- Strong domain familiarity
- Direct control over development
- Alignment with internal product vision
However:
- Limited experience with IEC 62304 and FDA compliance
- Slower ramp-up for specialized engineering
- Increased risk of architectural misalignment
Partnering with a Specialized Engineering Firm
- Access to medical device software engineering services
- Established frameworks for compliance and validation
- Faster path to FDA-ready diagnostic platforms
A Proven Approach to Engineering Diagnostic Medical Device Software
Building diagnostic systems requires more than talent, it requires a structured, repeatable engineering methodology aligned with regulatory and clinical demands.
High-performing teams follow a lifecycle that integrates compliance, architecture, and validation from the start.
1. Regulatory & Clinical Discovery
- Define intended use and clinical workflows
- Identify regulatory classification (FDA, SaMD)
- Map risks using ISO 14971
2. System Architecture & Design
- Design clinical-grade diagnostic software architecture
- Define data pipelines and processing layers
- Establish interoperability (FHIR, HL7)
3. Engineering & Integration
- Develop modular, traceable systems
- Implement real-time data processing
- Integrate AI models where applicable
4. Verification, Validation & Compliance
- Execute structured V&V processes
- Ensure full traceability across system components
- Prepare documentation for FDA submission
5. Deployment & Post-Market Monitoring
- Monitor system performance in real-world use
- Maintain compliance with regulatory updates
- Continuously improve diagnostic accuracy
Engineering Lifecycle vs Traditional Development
Phase | Diagnostic Software Engineering | Traditional Development |
Discovery | Regulatory + clinical driven | Business driven |
Design | Risk-based architecture | Feature-based design |
Development | Traceable, controlled | Agile, flexible |
Testing | Clinical + regulatory validation | Functional testing |
Deployment | Monitored, regulated | Continuous release |
A structured, compliance-driven approach is what transforms software into a clinically reliable, regulatory-approved diagnostic system.
Future of Diagnostic Software: Edge AI, Real-Time Decisioning, and Connected Care Ecosystems
The future of diagnostic medical device software is being shaped by a convergence of edge AI, real-time decisioning, and fully connected healthcare ecosystems, fundamentally redefining how and where clinical insights are generated.
Instead of relying on centralized, delayed analysis, modern diagnostic systems are moving intelligence closer to the point of care, enabling on-device processing, instant interpretation of physiological signals, and immediate clinical feedback.
This shift not only reduces latency but also enhances reliability in critical use cases where every second matters. At the same time, diagnostic platforms are evolving into interoperable systems that seamlessly integrate with EHRs, remote monitoring tools, and broader digital health infrastructures through standards like FHIR and HL7.
Combined with advancements in AI-driven analytics and continuous data streams, this creates a new paradigm where diagnostics are no longer episodic but continuously adaptive, delivering predictive insights, improving clinical workflows, and ultimately enabling more proactive, data-driven patient care.
Why CitrusBits for Diagnostic Medical Device Software Engineering
Building diagnostic software is not just about writing code, it’s about engineering systems that can withstand regulatory scrutiny, deliver clinical accuracy, and scale in complex healthcare environments.
This is where most development teams fall short.
CitrusBits operates at the intersection of AI, XR, and regulated healthcare engineering, enabling organizations to move from concept to FDA-ready diagnostic systems with confidence.
If you’re building or scaling a diagnostic platform and need:
- FDA-ready diagnostic software engineering
- Expertise in IEC 62304 and regulated system design
- Scalable, real-time, AI-powered architectures
Then it’s time to work with a partner that understands both technology and regulation at a system level.
Table of Contents
1) What Defines Diagnostic Medical Device Software Engineering?
2) Regulatory-First Architecture: Designing for FDA, IEC 62304, and ISO 14971
3) Architectural Implications
4) System Architecture for Diagnostic Platforms: Designing for Real-Time, Clinical-Grade Performance
5) Core Architectural Components
6) AI in Diagnostic Systems: From Algorithms to FDA-Validated Intelligence
7) Software Verification, Validation (V&V), and Clinical Readiness
8) Hidden Risks in Diagnostic Software Development (Why Most Projects Fail)
9) Build vs Partner: When to Outsource Diagnostic Software Engineering
10) A Proven Approach to Engineering Diagnostic Medical Device Software
11) Engineering Lifecycle vs Traditional Development
12) Future of Diagnostic Software: Edge AI, Real-Time Decisioning, and Connected Care Ecosystems
13) Why CitrusBits for Diagnostic Medical Device Software Engineering
Innovate the Future of Health Tech
CitrusBits helps MedTech leaders build smarter apps, connected devices, and XR health solutions that truly make an impact.
